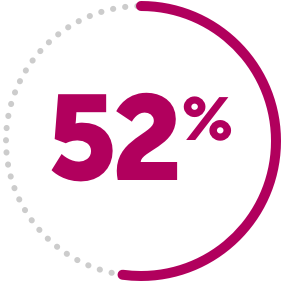
IMPORTANT SAFETY INFORMATION
ABOUT HUMIRA® (adalimumab)1
What is the most important information I should know about HUMIRA?
You should discuss the potential benefits and risks of HUMIRA with your doctor. HUMIRA is a TNF blocker medicine that can lower the ability of your immune system to fight infections. You should not start taking HUMIRA if you have any kind of infection unless your doctor says it is okay.
- Serious infections have happened in people taking HUMIRA. These serious infections include tuberculosis (TB) and infections caused by viruses, fungi, or bacteria that have spread throughout the body. Some people have died from these infections. Your doctor should test you for TB before starting HUMIRA, and check you closely for signs and symptoms of TB during treatment with HUMIRA, even if your TB test was negative. If your doctor feels you are at risk, you may be treated with medicine for TB.
- Cancer. For children and adults taking TNF blockers, including HUMIRA, the chance of getting lymphoma or other cancers may increase. There have been cases of unusual cancers in children, teenagers, and young adults using TNF blockers. Some people have developed a rare type of cancer called hepatosplenic T-cell lymphoma. This type of cancer often results in death. If using TNF blockers including HUMIRA, your chance of getting two types of skin cancer (basal cell and squamous cell) may increase. These types are generally not life-threatening if treated; tell your doctor if you have a bump or open sore that doesn’t heal.
What should I tell my doctor BEFORE starting HUMIRA?
Tell your doctor about all of your health conditions, including if you:
- Have an infection, are being treated for infection, or have symptoms of an infection
- Get a lot of infections or infections that keep coming back
- Have diabetes
- Have TB or have been in close contact with someone with TB, or were born in, lived in, or traveled where there is more risk for getting TB
- Live or have lived in an area (such as the Ohio and Mississippi River valleys) where there is an increased risk for getting certain kinds of fungal infections, such as histoplasmosis, coccidioidomycosis, or blastomycosis. These infections may happen or become more severe if you use HUMIRA. Ask your doctor if you are unsure if you have lived in these areas
- Have or have had hepatitis B
- Are scheduled for major surgery
- Have or have had cancer
- Have numbness or tingling or a nervous system disease such as multiple sclerosis or Guillain-Barré syndrome
- Have or had heart failure
- Have recently received or are scheduled to receive a vaccine. HUMIRA patients may receive vaccines, except for live vaccines. Children should be brought up to date on all vaccines before starting HUMIRA
- Are allergic to rubber, latex, or any HUMIRA ingredients
- Are pregnant, planning to become pregnant, breastfeeding, or planning to breastfeed
- Have a baby and you were using HUMIRA during your pregnancy. Tell your baby’s doctor before your baby receives any vaccines
Also tell your doctor about all the medicines you take. You should not take HUMIRA with ORENCIA® (abatacept), KINERET® (anakinra), REMICADE® (infliximab), ENBREL® (etanercept), CIMZIA® (certolizumab pegol), or SIMPONI® (golimumab). Tell your doctor if you have ever used RITUXAN® (rituximab), IMURAN® (azathioprine), or PURINETHOL® (mercaptopurine, 6-MP).
What should I watch for AFTER starting HUMIRA?
HUMIRA can cause serious side effects, including:
- Serious infections. These include TB and infections caused by viruses, fungi, or bacteria. Symptoms related to TB include a cough, low-grade fever, weight loss, or loss of body fat and muscle.
- Hepatitis B infection in carriers of the virus. Symptoms include muscle aches, feeling very tired, dark urine, skin or eyes that look yellow, little or no appetite, vomiting, clay-colored bowel movements, fever, chills, stomach discomfort, and skin rash.
- Allergic reactions. Symptoms of a serious allergic reaction include hives, trouble breathing, and swelling of your face, eyes, lips, or mouth.
- Nervous system problems. Signs and symptoms include numbness or tingling, problems with your vision, weakness in your arms or legs, and dizziness.
- Blood problems (decreased blood cells that help fight infections or stop bleeding). Symptoms include a fever that does not go away, bruising or bleeding very easily, or looking very pale.
- Heart failure (new or worsening). Symptoms include shortness of breath, swelling of your ankles or feet, and sudden weight gain.
- Immune reactions including a lupus-like syndrome. Symptoms include chest discomfort or pain that does not go away, shortness of breath, joint pain, or rash on your cheeks or arms that gets worse in the sun.
- Liver problems. Symptoms include feeling very tired, skin or eyes that look yellow, poor appetite or vomiting, and pain on the right side of your stomach (abdomen). These problems can lead to liver failure and death.
- Psoriasis (new or worsening). Symptoms include red scaly patches or raised bumps that are filled with pus.
Call your doctor or get medical care right away if you develop any of the above symptoms.
Common side effects of HUMIRA include injection site reactions (pain, redness, rash, swelling, itching, or bruising), upper respiratory infections (sinus infections), headaches, rash, and nausea. These are not all of the possible side effects with HUMIRA. Tell your doctor if you have any side effect that bothers you or that does not go away.
Remember, tell your doctor right away if you have an infection or symptoms of an infection, including:
- Fever, sweats, or chills
- Muscle aches
- Cough
- Shortness of breath
- Blood in phlegm
- Weight loss
- Warm, red, or painful skin or sores on your body
- Diarrhea or stomach pain
- Burning when you urinate
- Urinating more often than normal
- Feeling very tired
HUMIRA is given by injection under the skin.
This is the most important information to know about HUMIRA. For more information, talk to your health care provider.
Uses
HUMIRA is a prescription medicine used:
- To reduce the signs and symptoms of:
- Moderate to severe rheumatoid arthritis (RA) in adults. HUMIRA can be used alone, with methotrexate, or with certain other medicines. HUMIRA may prevent further damage to your bones and joints and may help your ability to perform daily activities.
- Moderate to severe polyarticular juvenile idiopathic arthritis (JIA) in children 2 years of age and older. HUMIRA can be used alone or with methotrexate.
- Psoriatic arthritis (PsA) in adults. HUMIRA can be used alone or with certain other medicines. HUMIRA may prevent further damage to your bones and joints and may help your ability to perform daily activities.
- Ankylosing spondylitis (AS) in adults.
- Moderate to severe hidradenitis suppurativa (HS) in people 12 years and older.
- To treat moderate to severe Crohn’s disease (CD) in adults and children 6 years of age and older.
- To treat moderate to severe ulcerative colitis (UC) in adults and children 5 years of age and older. It is not known if HUMIRA is effective in people who stopped responding to or could not tolerate anti-TNF medicines.
- To treat moderate to severe chronic plaque psoriasis (Ps) in adults who are ready for systemic therapy or phototherapy, and are under the care of a doctor who will decide if other systemic therapies are less appropriate.
- To treat non-infectious intermediate (middle part of the eye), posterior (back of the eye), and panuveitis (all parts of the eye) in adults and children 2 years of age and older.
US-HUM-210186